Hexaxim Vaccine - Childhood immunisation
RM197.00
RM227.00
Category: Children’s Health
Quantity: In Stock
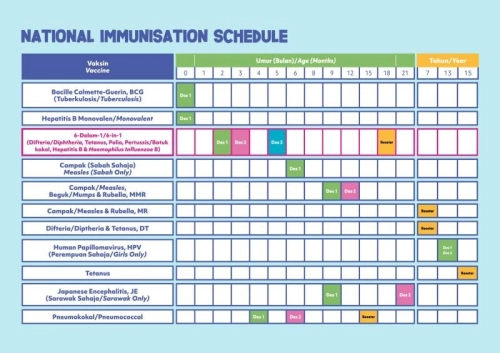
Hexaxim vaccine is included in the childhood immunisation schedule of several countries, including Malaysia, where it is part of the Malaysia National Immunisation Programme (NIP).
Indications/Uses
Hexaxim (DTaP-IPV-HB-Hib) is indicated for primary and booster vaccination of infants and toddlers from six weeks to 24 months of age against diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis and invasive diseases caused by Haemophilus influenzae type b (Hib).
The primary vaccination consists of 3 doses of 0.5 ml to be administered at intervals of at least four weeks and as per schedules 6, 10, 14 weeks; 2, 3, 4 months; 3, 4, 5 months; 2, 4, 6 months.
All vaccination schedules including the WHO Expanded Program on Immunisation (EPI) at 6, 10, 14 weeks of age can be used whether or not a dose of hepatitis B vaccine has been given at birth.
Where a dose of hepatitis B vaccine is given at birth: Hexaxim can be used for supplementary doses of hepatitis B vaccine from the age of 6 weeks.
Hexaxim can be administered simultaneously with a pneumococcal polysaccharide conjugate vaccine, measles, mumps, rubella (MMR) containing vaccines, rotavirus vaccines, a meningococcal C conjugate vaccine or a meningococcal group A, C, W-135 and Y conjugate vaccine, as no clinically relevant interference in the antibody response to each of the antigens have been shown.
Limited stock - Vaccine availability operates on a first-come-first-serve basis, and to secure your vaccine, we accept a non-refundable deposit.
Please Click Here to view Malaysia Childhood National Immunisation Programme (NIP).
Inquiry - Hexaxim Vaccine - Childhood immunisation